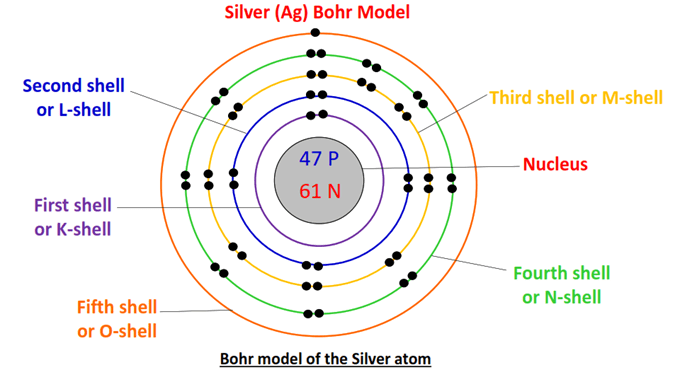
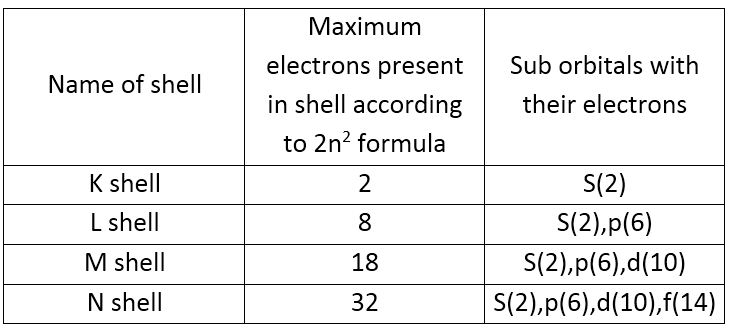
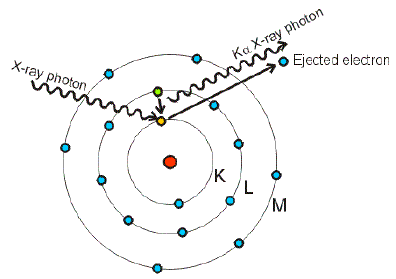
What is the difference between Shell , Subshell and Orbital – Digital Kemistry - Best Online Free Chemistry Learning

Shell - Electron - Character, Definition, subshell, energy of subshell, Arrangement of electrons in shell with FAQs

m shell on an atom has 3 electrons write the electronic configurationfind atomic number draw bohr - Brainly.in
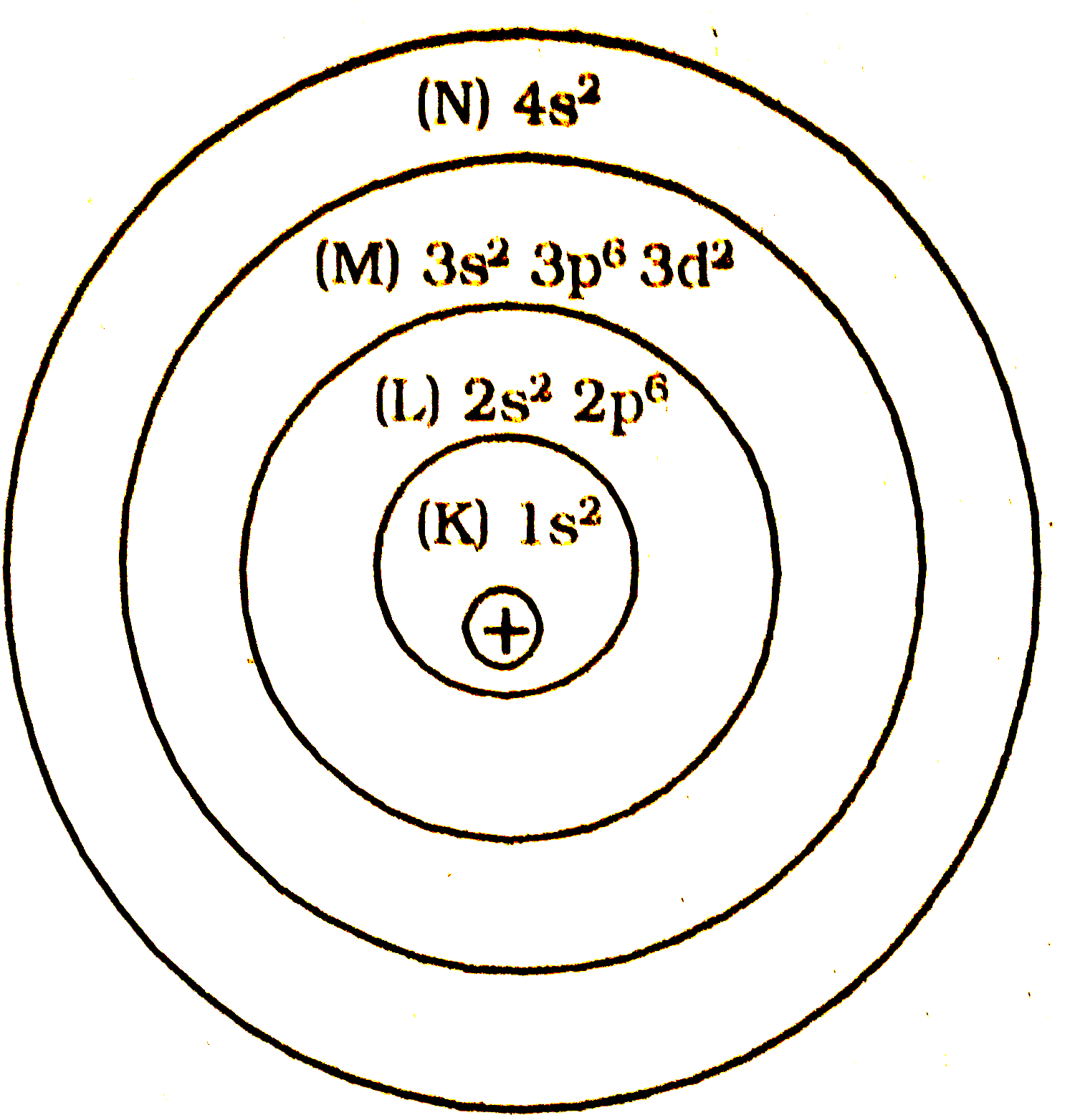
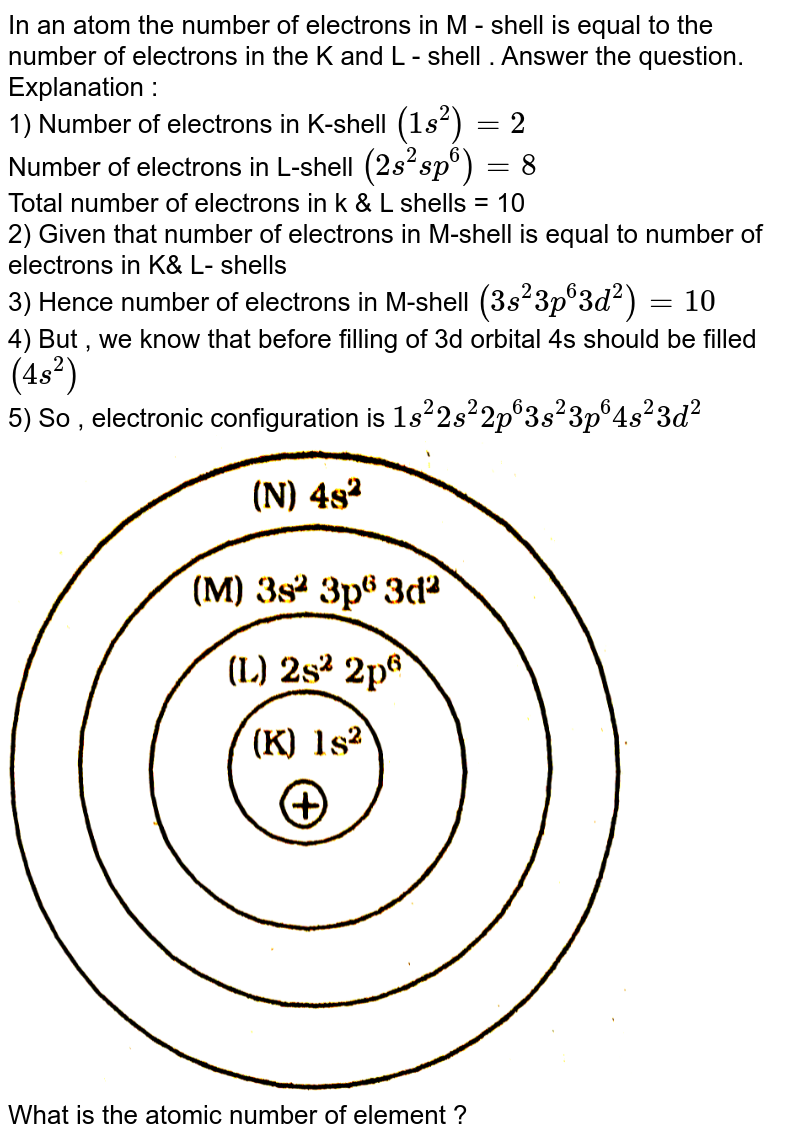
In an atom the number of electrons in M - shell is equal to the number of electrons in the K and L - shell . Answer the question. Explanation : 1)

If K and Lshells of an atom are full and in Mshell there is only one electron then what would be the total number of electrons in the atom Name the element
From amongst K, L, M and N shells, which is associated with lowest energy and which one is associated with highest energy and why? - Quora

The capacity of M shell is 18 , then why does M shell of calcium has 8 electrons though it's atomic number is 20
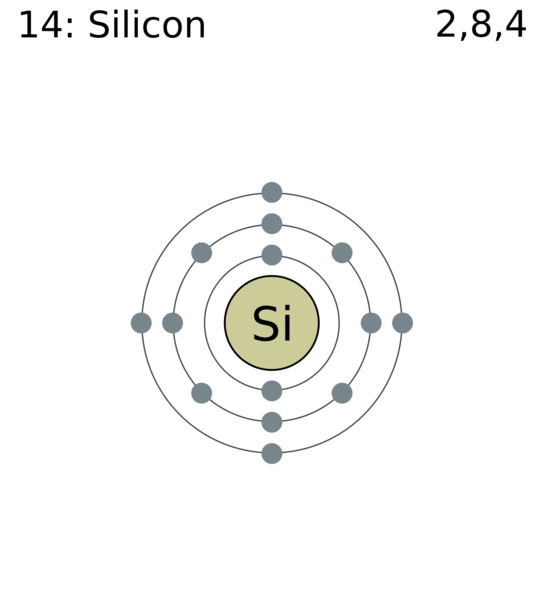
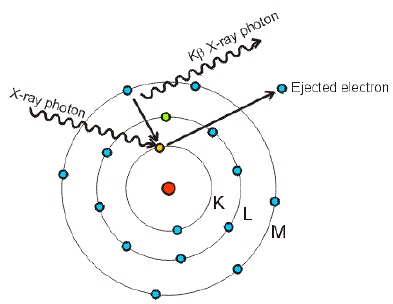
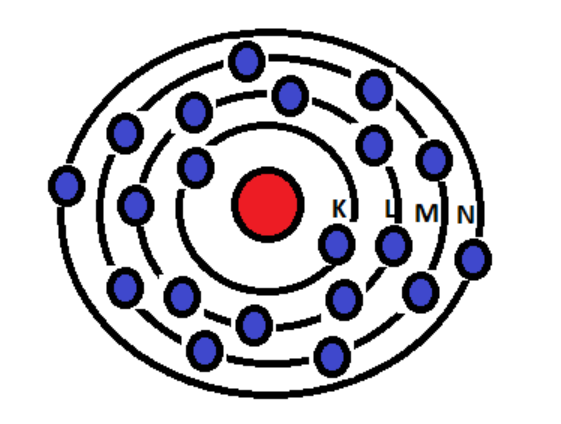

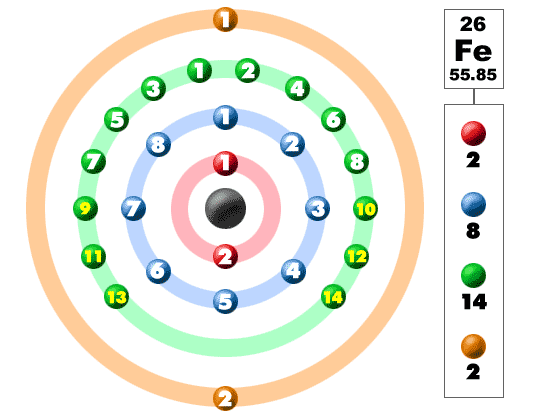


![Solved] How many electrons are present in M - Shell of an element wi Solved] How many electrons are present in M - Shell of an element wi](https://storage.googleapis.com/tb-img/production/22/03/F1_Shraddha_Neha%20G_17.12.2021_D1_Corrected%201.png)