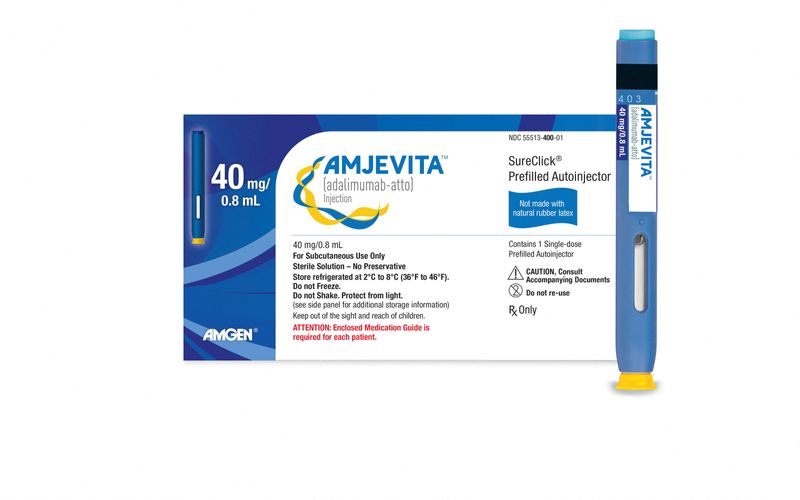
Humira von AbbVie bekommt einen US-Konkurrenten, aber die Kosten könnten hoch bleiben | MarketScreener

PDF) The Expiry of Humira® Market Exclusivity and the Entry of Adalimumab Biosimilars in Europe: An Overview of Pricing and National Policy Measures

The adalimumab (HUMIRA ® ) Pen. Reprinted with permission from Clinical... | Download Scientific Diagram